The first step in electronics is understanding the concepts of current and voltage. There is a hydraulic analogy that helps to understand electricity, but here we will try to cover how it works on atomic and electron level.
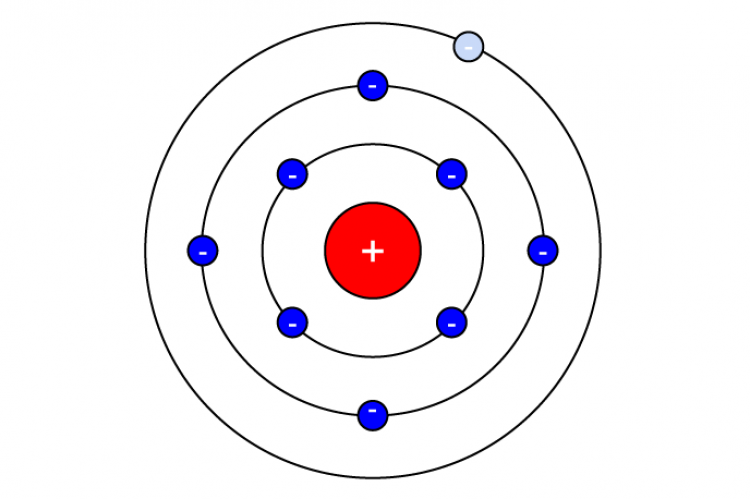
Atoms have a core which consists of protons and neutrons making it positively charged. Atoms have shells with electrons around the core. The outer shell (also called valence shell) houses free electrons and they can move freely from atom to atom. Materials that have free electrons can conduct electricity. The drawing above shows a Bohr atom model with one free electron in the valance shell.
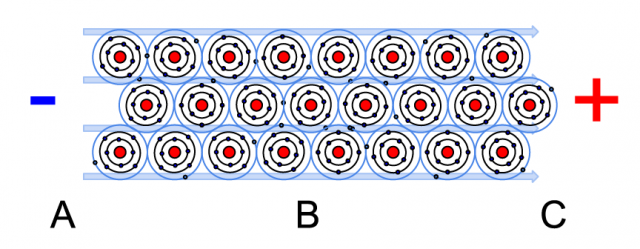
In a wire all the atoms are connected together and when charge is applied on the material we can influence the flow of free electrons. The movement of these electrons is called current. The amount of electrons that flows, for example at point B, is measured per time unit. If over 6 quintillion (6 with 18 zeros) of electrons will pass at that point during one second we call it 1 ampere (A). The difference in charge between points A and C is called potential. The potential determines the force that the electrons move through the conductor. It is voltage.
Imagine a ball that is rolling down the hill. The slope of the hill can be called electrical current. The larger the difference in height (voltage) between point A and C the faster the ball (electron) will roll. You can measure the voltage (V) between two points just as you can measure the height between the top and the bottom of the hill.

In the drawing with the electrons you can see that charge is flowing from the negative to the positive pole. However we say that the current flows opposite to the charge flow. This has historical reasons and in that time they defined the flow using the ions which were positively charged. In practice it makes no difference how you see it, as long as you keep it constant with what is positive and what is negative.